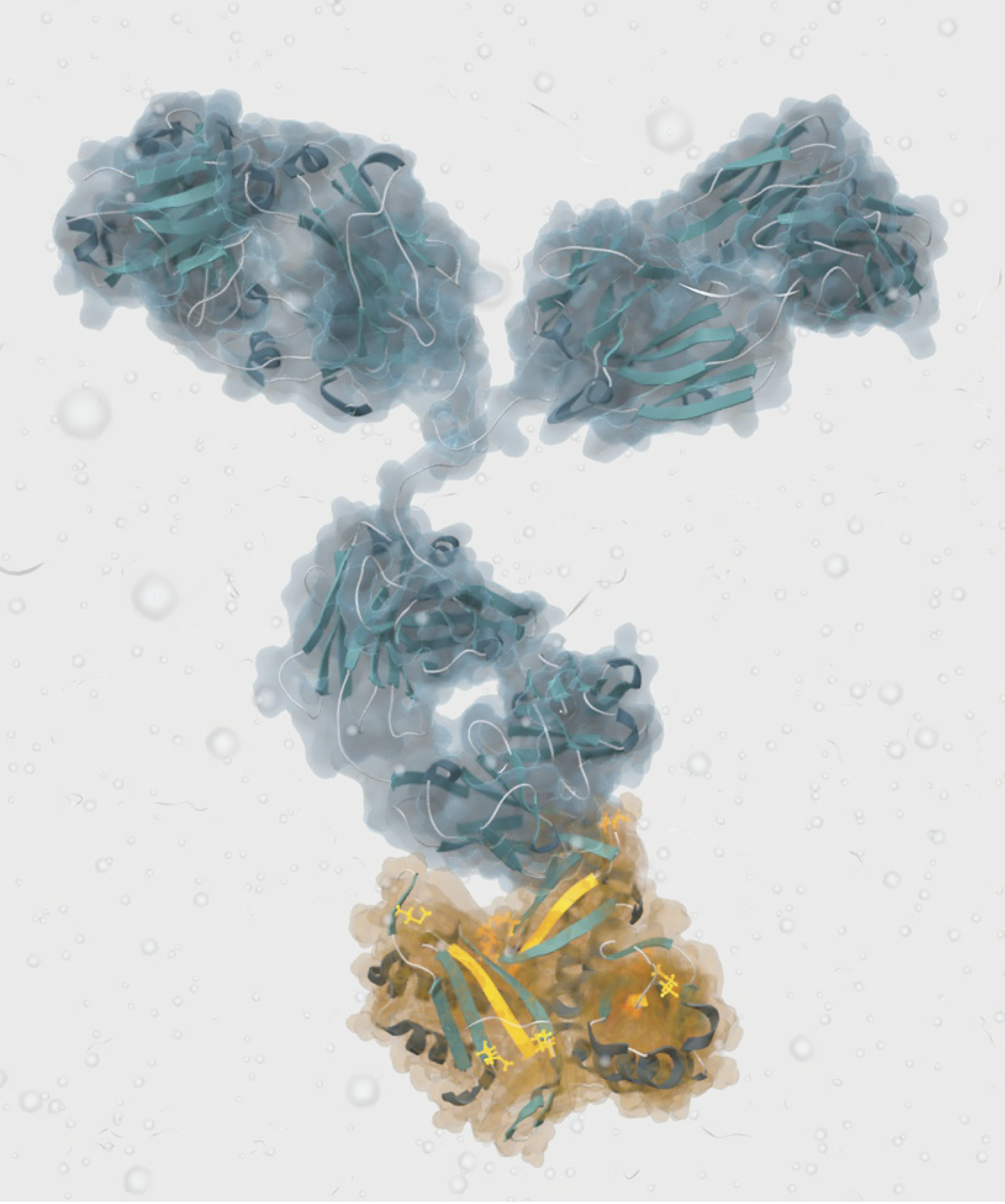
At the forefront of what we are doing are our antibody engineering capabilities to design novel molecules that address inflammation and cancer and exhibit best-in-class profiles for safety and efficacy aimed at effectively improving patient outcomes and quality of life.
OncoOne Antibody Technology
Engineering for Precision Therapeutics
The Pioneers for oxMIF and Wizards of Antibody Engineering are at the forefront of precision medicine. Monoclonal antibodies have revolutionized immunotherapy and are the preferred modalities to target cytokines and tumor antigens. Antibody engineering has emerged as a critical step to optimize their therapeutic potential. At OncoOne, our world-class antibody engineers develop and optimize unique, tailor-engineered mAbs with precisely defined functionalities, specifically tailored for individual therapeutic indications.
OncoOne is pioneering two cutting-edge technology platforms that are unlocking a new era of therapeutic breakthroughs. These transformative platforms, driven by
antibody engineering, are poised to revolutionize the treatment of major
chronic inflammatory and oncological diseases.

READ NEXT
OncoOne's Technology Platforms Anti-oxMIF and PreTarg-it ®
Two new ground-breaking platforms to deliver innovative treatments with unmatched safety and efficacy, surpassing mere symptom relief and making significant contributions to a healthier global community